The modern slavery reporting platform for the medical research sector
The modern slavery reporting platform for the medical research sector
Manage complex research supply chains, streamline supplier due diligence, and collaborate with industry peers through a shared platform built for the sector.
Trusted by leading medical research institutes


















Modern slavery reporting is becoming more complex across research supply chains
Medical research institutes operate across global, highly specialised supply chains spanning laboratory equipment, clinical trials, pharmaceuticals, and outsourced services.
Procurement, ESG, and governance teams must collect reliable supplier disclosures and assess risks across international vendors, research partners, and service providers.
Institutes must also produce clear, defensible reporting aligned to regulatory requirements, ethical research frameworks, and expectations from funders, partners, and stakeholders.
No organisation can solve these challenges alone.
Why the Medical Research platform exists
Shared risks require shared solutions
Medical research supply chains are global, specialised, and interconnected. From laboratory materials and clinical trials to outsourced services and international collaborations, modern slavery risks are often embedded in complex, multi-tier networks.
Medical Research Institutes
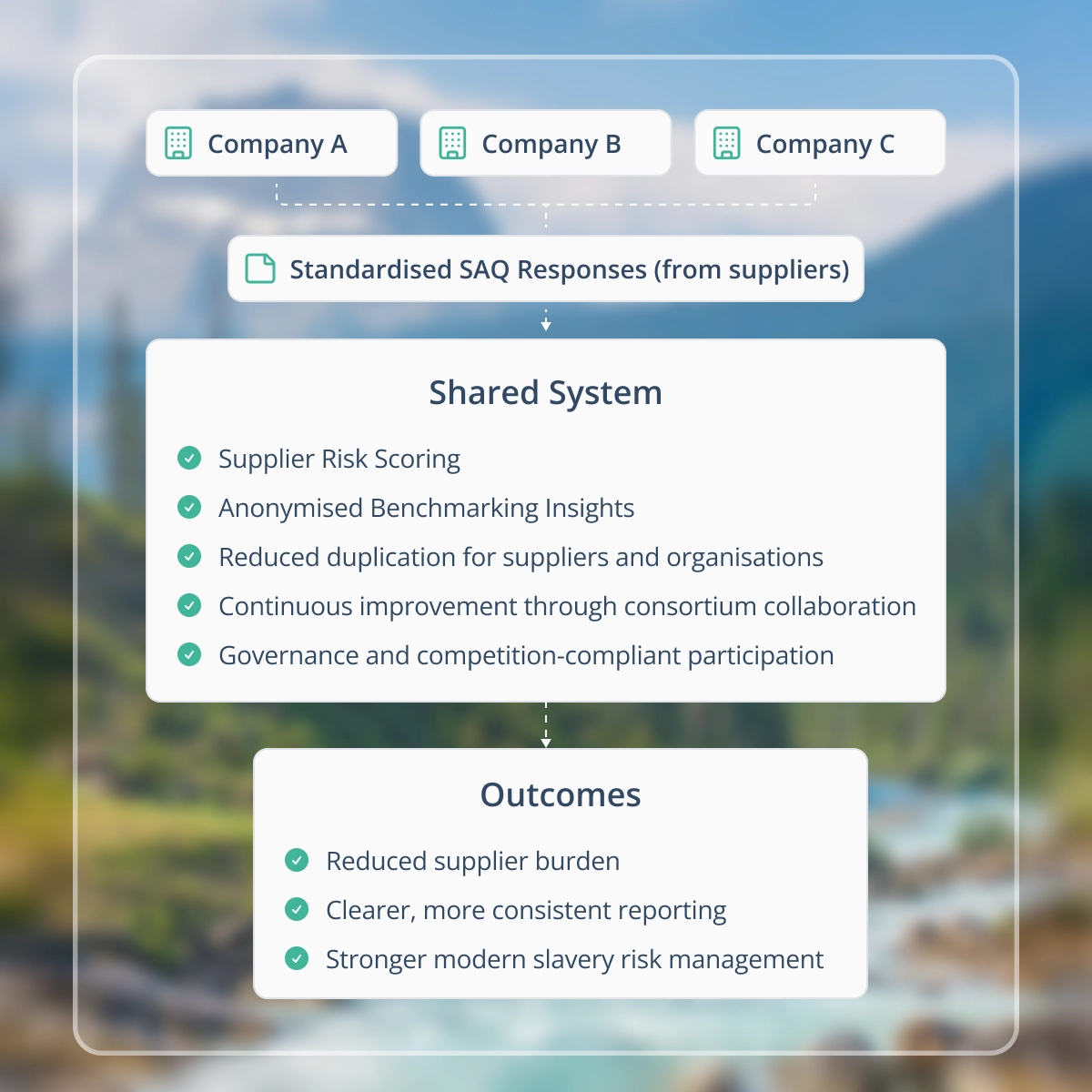
The sector now works together through the platform to:
- Use one shared Modern Slavery Self-Assessment Questionnaire (SAQ) to reduce duplication
- Lift capability across the industry through shared learning and regular collaboration opportunities
- Deliver stronger evidence for internal and external reporting with consistently better supplier disclosures
Sector impact
Industry collaboration delivering measurable impact
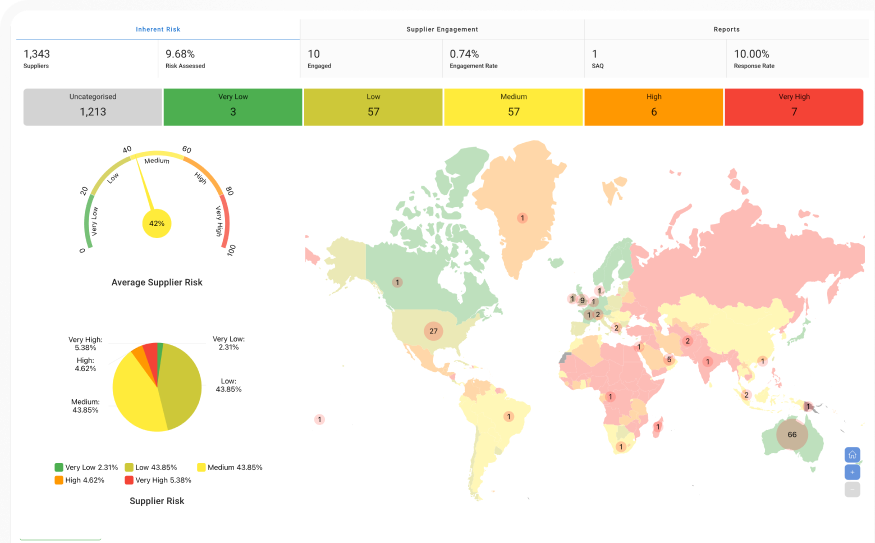
Suppliers onboarded across the platform
Supplier engagement rate
Medical research institutes participating
How the medical research platform works
Collect, assess, and report on modern slavery risk through one connected platform built for compliance, transparency, and continuous improvement.
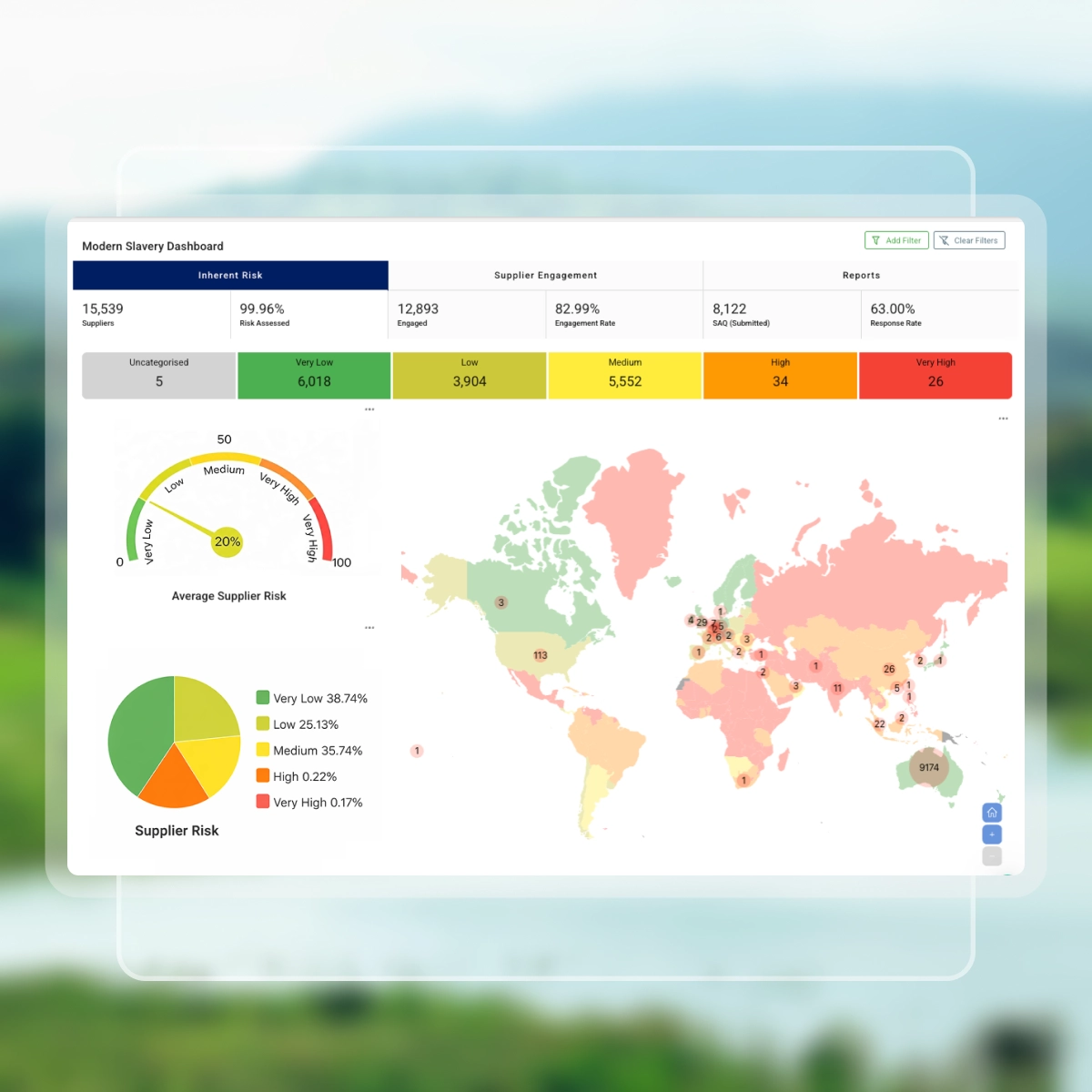
Establish your baseline
Upload your supplier list to instantly generate country and industry-based inherent risk ratings, giving you a clear baseline of modern slavery risk across your supply chain before deeper assessments begin.
Ratings informed by trusted data sources including:
- Global Slavery Index
- Corruption Perceptions Index
- Global Peace Index
- Social Progress Index
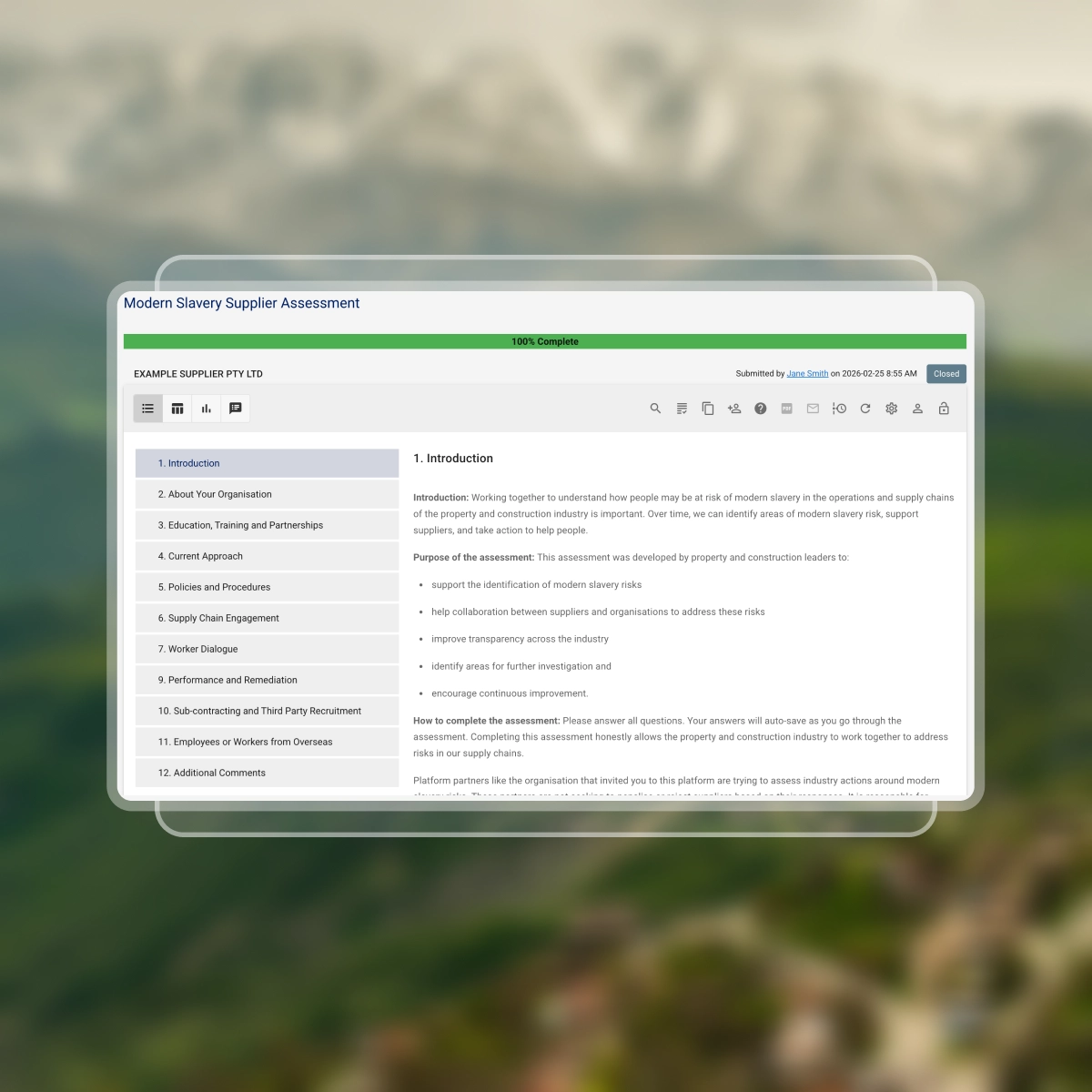
Engage suppliers with the shared SAQ
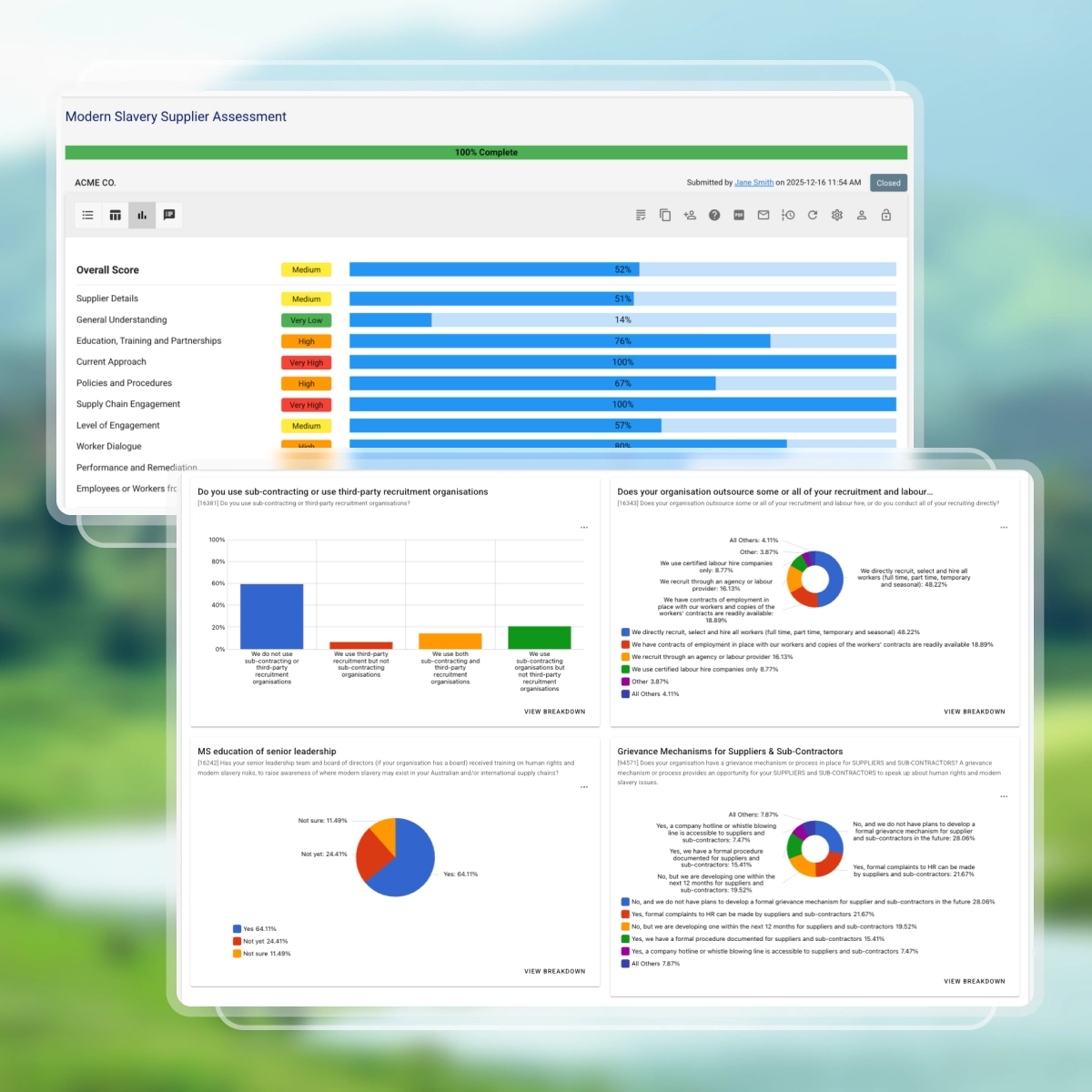
Assess modern slavery risk across key supplier practices:
- Organisation profile, including structure, workforce, and geographic footprint
- Labour practices and human rights risks, including recruitment, employment conditions, and overseas workers
- Supply chain arrangements, including sourcing of materials, equipment, and services
- Governance and risk management, including policies, due diligence, and assessment processes
- Worker protections and remediation, including grievance mechanisms and risk responses
- Training and continuous improvement to strengthen supplier capability
Suppliers only see questions relevant to them through conditional logic.
Report & Improve
Automatically score SAQ responses to assess residual supplier, human rights, and ESG risk using configurable models.
Review results alongside inherent risk and external intelligence to prioritise high-risk suppliers. Interactive dashboards provide shared visibility across procurement, risk, and leadership teams.
Bring together internal ESG and supplier data, including spend, risk ratings, and assessments, in one secure platform with integrations to trusted data providers such as Dow Jones, Dun & Bradstreet, and Sedex.


Move beyond reactive reporting to strengthen governance, improve supplier accountability, and embed ESG performance into decision-making.
When organisations act independently across the sector
These issues can arise:
- Reporting becomes repetitive and low value
- Risk scoring varies widely
- Duplication becomes entrenched
- Suppliers receive multiple, inconsistent requests
- Effort increases without better outcomes
A shared, sector-wide platform reduces duplication, improves consistency, and strengthens modern slavery risk reporting.


Collaboration advantage
A sector-wide working group aligned to AAMRI
The Informed 365 Medical Research Collaboration brings together leading institutes to address modern slavery risks collectively, and is supported by expert facilitation from Robin Mellon, CEO of Better Sydney.
The collaboration includes:
- Regular cross-institute forums and knowledge sharing
- Shared analysis of aggregated supplier data and emerging risks
- Ongoing access to curated learning resources
- Continuous refinement of the shared SAQ to reflect the evolving market.
This collaboration ensures the platform reflects real research supply chain challenges and evolving regulatory expectations.

Informed 365 launched the platform in partnership with the Australian Association of Medical Research Institutes in 2025.
The platform standardises how institutes identify, assess, and report on modern slavery risks across global research supply chains, improving consistency, comparability, and reporting quality
Designed for organisations across the medical research sector, including:
- Independent medical research institutes
- Institute-led and hospital-affiliated research organisations
- Clinical and translational research institutes
- Specialised biomedical and laboratory research organisations
- And more
Client Testimonials

“By working together as a unified AAMRI membership base, we can create a more efficient and effective approach to managing Modern Slavery compliance. This collaboration enables data sharing and delivers substantial benefits to participating suppliers, ultimately contributing to the broader goal of eradicating Modern Slavery within both domestic and international supply chains.”

“Informed 365 has been a game-changer for the Institute. Its intuitive platform has streamlined our decision-making and compliance processes. We can easily track progress, identify risks and work with those high-risk suppliers throughout our supply chain to make a real impact.”

Platform impact
Visible uplift across the sector:
- Suppliers reusing their SAQ across multiple institutes
- Thousands of data points mapped across governance, labour, and supply chain controls
- Higher supplier completion rates, with more consistent reporting across the sector
- Improved supplier capability through access to shared learning resources
See the platform in action
See how your organisation can streamline reporting, strengthen due diligence, and participate in industry collaboration.